|
Integrated Concurrent Exercise Training
In Minnesota, concurrent enrollment courses are college courses offered at the high school, usually taught by a trained high school teacher. These are offered in partnership with a college or university. Students who successfully complete these courses generate both high school and transcripted college credit from the partnering postsecondary institution. Many people refer to these courses as College in the High School. There is no cost to the student to participate in these courses. Minnesota’s concurrent enrollment programs offer thousands of Minnesota students access to rigorous college courses in their high school buildings. Research shows that high school students who participate in an accelerated learning option, such as concurrent enrollment, benefit greatly from: • Exposure to high expectations. By participating in concurrent enrollment, high school students complete college requirements that allow for greater flexibility when they enter the university setting full-time. Many concurrent enrollment alums find they are able to pursue second majors, participate in study abroad opportunities, and internships. Not only do concurrent enrollment students get a step ahead of other entering freshman in terms of credits, but they also gain college-level skills from concurrent enrollment courses. Our courses challenge high school students to think critically, write academically, and read analytically, preparing students for greater success in college. Participating high schools also reap the benefits of concurrent enrollment. Partnerships developed between university faculty and high school teachers provide learning and training opportunities that may not otherwise exist. Additionally, concurrent enrollment students stay at the high school instead of leaving to attend classes at a university. Finally, through participation in concurrent enrollment, high schools establish themselves as education leaders by setting high standards, providing outstanding offerings, and preparing students for the 21st century.
Access to Rigorous College Courses in Your High School!
Related MDE resources:Related offsite resources:Concurrent training, the combination of divergent training stimuli such as a heavy resistance training (>70% 1RM) coupled with an endurance style of training (long, slow jogging) has received a large amount of skepticism over the years. Primarily, it is frequently postulated that the adaptations brought about through the utilization of resistance training will be inhibited by those of aerobic training and vice versa. A number of studies in the past have looked at this and, when looking at the outcome of these studies, one would conclude that these two styles of training do have inhibitory roles upon each other. However, much of the more recent studies questions this line of thinking. This being so, this article will present the evidence and let the reader decide for him/herself. The primary argument against the combination of the two divergent training interventions (aerobic training and resistance training) together in one training session is the, what was once thought, antagonistic molecular responses. Specifically, the increased signaling protein mechanistic target of rapamycin complex 1 (mTORC1) response following resistance training being inhibited via the increase of signaling protein AMP-activated protein kinase (AMPK) following an aerobic training stimulus. The reader may have come across these abbreviations (AMPK/mTOR) before, however, a brief description of these two signaling pathways will prove necessary. Exercise-Induced Signaling ProteinsmTOR has been coined as the “master regulator” within the muscle fiber concerning protein synthesis. So, regardless of where the signal may be coming from (amino acids/growth factors/stretch) the signal will inevitably lead to an increase in mTOR activity. This increase in mTOR activity can then lead to a myriad of signals but, concerning protein synthesis, its functions primarily alter transcription and translation factors. Increased activation of these factors allows for specific expression of various sequences on the DNA allowing for mRNA to be created. Once this mRNA is created, it is taken to the ribosome within the cytoplasm where it is ‘translated’ into new protein, thus, protein synthesis. Increased activation of mTOR allows this increased protein synthesis to take place. This being the case, mTOR is anabolic in nature. Now, when looking at AMPK, its role may seem opposite to that of mTOR. AMPK is an energy sensor of the muscle fiber. When ATP is rapidly hydrolyzed during intensive training, the AMP:ATP ratio gradually increases leading to increased activity of AMPK. AMPK, similar to mTOR, can then take part in a number of intracellular signaling processes. A few, but not all, of the pathways which AMPK plays a part in is mitochondrial biogenesis, increased PGC1alpha (a transcriptional co-factor leading to increased activity of mitochondrial genes encoded in the DNA), de-phosphorylation of FOXO which allows it to translocate to the nucleus and activate MURF-1 and Atrogin-1 (both proteolytic factors). This being the case, when concerning protein synthesis, we may conclude that AMPK is catabolic in nature. So, making the above paragraph less confusing, AMPK increases the oxidative properties of the given muscle fiber along with increasing the activity of proteolytic functions (opposite of protein synthesis) whereas mTOR increases activity of transcription and translation factors which enhance protein synthesis. Looking at this, it would seem rather evident then that AMPK and mTOR play opposite roles. For example, if AMPK is increased then mTOR is decreased and vice versa. And for the longest time, this was used as the reasoning to discredit concurrent training. However, much research has shown both an increase in mTOR and AMPK following a given training intervention with no decrease to mTOR or altered effects to protein synthesis. This may be due to an overshadowing effect of mTOR on AMPK or the acute time frame (relative to mTOR) that AMPK is increased. Much more research is needed in this area. However, the primary thing to understand is that, when looking at anabolic and catabolic signaling pathways, there is not an inverse relationship as was once thought. Concurrent Training: Past and Current ResearchOne of the primary studies which gave credence to the inadequacy between combined resistance and aerobic training is that of Hickson. In this study, Hickson has one group take part in resistance training five times a week, one group do aerobic training six times a week, and a third group take part in both the resistance and aerobic training. The training intervention for the resistance training consisted of, for all exercises, near-maximal loads. The endurance training group exercised at or near their VO2max. Again, the concurrent training group did both of these training interventions. Two things should be quite evident when looking at this information. For one, the overall frequency of training was significantly greater for the concurrent training group (11 total training sessions per week) than the other two groups. Further, each training session consisted of incredibly rigorous exercise. So, the key finding from these researchers may very well be due to over-reaching/overtraining and not inadequacy of these two divergent training stimuli. This is a very interesting study because, while I feel it does not discredit concurrent training, it does demonstrate that if concurrent training is inappropriately programmed, it can have negative effects on the desired adaptations. Stressors on the body work synergistically and the body has a finite capacity in its ability to adapt to said stressors. Thus, proper programming is paramount. Etv serials in telugu youtube. Looking at more recent research, Lundberg and colleagues have shown an increased anabolic effect following concurrent training as compared to either resistance training or aerobic training alone. However, when looking at the neural output of the individual (such as power output) there was shown to be a decrease in the concurrent training group as compared to the resistance-only trained group. The key issue here is the time in-between the two training interventions. For example, one of their studies did an aerobic training session with a 15-minute break and then a resistance training session. The power output decreased in this concurrent-trained group. However, the same researchers did another study which substituted the 15 minutes for 6 hours. In this case, there were no differences in power output with the concurrent trained group as compared to the resistance-only trained group. This being the case, time looks to be a key factor when enhanced neural adaptations are sought. Timing: The Key VariableIt must be noted that this article is specifically looking at intracellular signaling pathways leading to increased muscle size (hypertrophy). Thus, while much of the literature has found little evidence to demonstrate an attenuated anabolic effect, there has been ample evidence showing attenuated kinetic aspects of the given exercise. Specifically, as previously mentioned, if there is minimal time between the endurance-based training and the resistance-based training, the neural output ability of the individual will be decreased. The above mentioned may prove troublesome for sports which require high levels of neural activity. This is frequently the case during the off-season of which the coach will often hold the “conditioning” session early in the morning and then have the “weight room” session following. However, as previously mentioned, this side-effect can be overcome. If a concurrent training program is sought and neural adaptations are the primary goal, the two training sessions (endurance and resistance) should be spaced with a minimum of six hours apart. If six hours is given, it was demonstrated in the above-mentioned paper that not only did neural output of the concurrent trained group increase in line with the resistance-only trained group, but that the concurrent trained group also had a greater increase in muscle size. This being the case, time is a key variable for concurrent training and will dictate the overall adaptation. So again, when looking at anabolic signaling pathways within the skeletal muscle fiber, all have one thing in common, they must all go through (from what we know so far) the mTOR pathway. On the other hand, it would seem plausible to conclude that any activity which inhibits mTOR signaling would be catabolic in nature and subsequently have an inverse relationship with mTOR. This can, to a certain extent, be seen with AMPK. I say, “to a certain extent” because the primary mechanism which inhibits mTOR is TSC2. TSC2 is activated by AMPK. However, through a strenuous training bout, it must be understood that there are a number of mechanisms which activate mTOR such as stretch, amino acid influx, growth factors, among others. This being the case, even if AMPK does inhibit mTOR to some extent, the overall activity of mTOR may still exceed any inhibitory AMPK signaling. Therefore, I feel it unwise to talk about AMPK and mTOR as having an inverse relationship. AMPK and mTOR may very well both be highly active at the same time as has been shown in several past studies. So, let’s look at some of the key areas which may cause an enhanced mTOR signaling and subsequent hypertrophying stimulus with concurrent training. Possible Mechanisms at PlayIt is still somewhat speculated as to what is at play leading to this increased anabolic response, however, there are some areas which stand out as leading candidates. One possible mechanism is cell swelling. Coupling an endurance-based and resistance-based training intervention can lead to significant metabolic stress within the given muscle fiber. This increase in intracellular metabolite accumulation has been speculated to alter cell-membrane structure leading to a mechanotransducing effect of mechanically sensitive ion channels. Along with this, integrins which span the muscle fiber membrane which physically attach the extracellular matrix to the intracellular environment have also been postulated to increase in activity following cell swelling. This increase in integrin activity has been shown to lead to greater activity of anabolic properties within the cell. Moreover, this cell swelling has been demonstrated to increase the transportation of amino acid influx to the muscle fiber subsequently enhancing mTOR activity and protein synthesis Further, an extreme increase in intracellular calcium content has been shown to be present following a concurrent training intervention. This increase in calcium content has been demonstrated to activate mTORC1 leading to increase in protein synthesis and subsequent hypertrophy. Additionally, when coupling these apparent divergent stimuli, an increase in reactive oxygen species (ROS) has been shown to occur which may also acutely increase mTOR signaling. The last mechanism which may play a key role in the increased anabolic effect of concurrent training can be demonstrated via the number of muscle contractions which takes place during the training intervention. It is, without question, greater in the concurrent training group as compared to resistance-only or endurance-only trained group. This is important because there are many signaling proteins which are located on the z-line of the sarcomere (the smallest functional unit of a muscle fiber). Two key signaling proteins which lie at the z-line of a sarcomere is alpha-actinin and phospholipase D (PLD).Looking at PLD, upon muscle contraction, PLD becomes active which can cause an increase in the production of phosphatidic acid. Phosphatidic acid has been shown to bind to mTOR leading to its activation and subsequent protein synthesis. Further, looking at alpha-actinin (a protein which binds actin filaments), upon muscle contraction it has been shown to increase in activity and subsequently causing an increase in calcineurin activity which, too, has been shown to have an anabolic effect. A Few CaveatsOf course, with anything, there are caveats. The first, and in my modest opinion, most important, is the necessity to specify the training stimulus to meet the athletes level of preparedness. This can be viewed as an inverse relationship. As the athlete becomes more and more advanced, the training needs to become more specific with less emphasis placed on general physical preparation in order to reap greater and more specific adaptations. We'd love for you to download a trial of SketchUp Pro. With the trial, you can run the full version SketchUp Pro, including features like LayOut and Style Builder. Sketchup pro license free 2018. Dec 5, 2013 - If you would like to download past versions of google sketchup free look at the list below! We also have links to download the latest. Free updates to the latest version of SketchUp Pro are emailed out during the 1-year license term. Please email your name, school, and serial number to. Need a SketchUp Pro Student License? Then you're in the right place. Get a full-featured, single-user license priced lower than the cost of a text book, at only. This denotes the importance of a proper periodized scheme in relation to the overall magnitude of the concurrent training stimulus. As the athlete increases in specialty, this warrants a more specific approach concerning the necessary biomotor, bioenergetic, and biodynamic peculiarities of his/her sport. The above being the case, year-round concurrent training is less than advantageous for a college American Football player as compared to the novice middle schooler. However, a question may be, when might be a good time for the more advanced athlete to utilize a concurrent training approach? Some good times to use a concurrent training approach would be during the training of novice and moderate-novice athletes. However, even for elite or sub-elite athletes, it may be advantageous to implement a concurrent style of training during the off-season, recovery/regeneration sessions or, as coined by the late Tudor Bompa, during the anatomical adaptation phase. I say this because the predominant positive effect elicited by concurrent training, at least for power sports, is at the molecular level. As previously mentioned, concurrent training increases protein synthesis via increased mTOR activity and subsequent increased downstream factors such as P70s6k, 4EBP1, and EIF2BE which then increases protein synthesis and overall muscle mass. Further, concurrent training has been shown to increase lactate activity which assists in tissue regeneration. And lastly, concurrent training has been demonstrated to increase mitochondrial biogenesis which enhances the athlete’s oxidative capabilities. Thus, while much of these adaptations may be non-specific for many sports, they can wisely be implemented in the off-season or recovery session to increase protein synthesis and subsequent hypertrophy, increase lactate activity to assist in tissue repair, and increase oxidative properties such as mitochondrial biogenesis as to further assist in the recovery process. ConclusionLastly, when taking a step away from signaling proteins and looking directly at the transcriptome (gene expression) following concurrent training as compared to resistance training, there are multiple sequences of DNA which alter both oxidative and muscle fiber contractile properties simultaneously that have been shown to be upregulated to a greater degree in the concurrent training group. This further demonstrates the necessity to view the complex intracellular system as a network which takes part in cross-talk rather than a linear signaling process which work in an orderly fashion. There are many variables which need to be taken into consideration when implementing a concurrent training plan. The athletes training state, current position within the individuals yearly plan, the individual's overall goal, the time in-between sessions, the total number of training interventions programmed, among others. However, taking the information given so far, does an aerobic session prior to a resistance training session automatically attenuate any possible anabolic effects from the resistance training intervention? No. Rather, you may actually have a greater anabolic effect with the two divergent training stimuli as compared to either aerobic or resistance training alone. So, give it a try. References
Luke Olsen is a current graduate student within the Exercise Physiology department at the University of Kansas with research interests related to exercise’s role in cellular and molecular physiology.
Loading Comments..
Integrated Concurrent Exercise Training
Published online 2014 Oct 30. doi: 10.1007/s40279-014-0252-0
PMID: 25355186
This article has been cited by other articles in PMC.
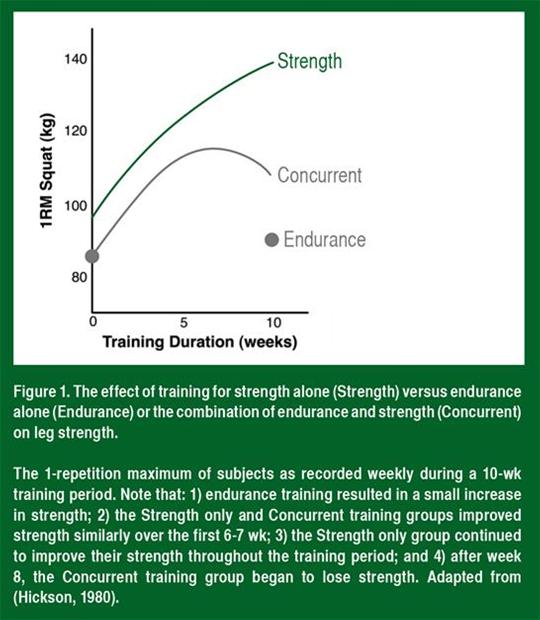
AbstractVery few sports use only endurance or strength. Outside of running long distances on a flat surface and power-lifting, practically all sports require some combination of endurance and strength. Endurance and strength can be developed simultaneously to some degree. However, the development of a high level of endurance seems to prohibit the development or maintenance of muscle mass and strength. This interaction between endurance and strength is called the concurrent training effect. This review specifically defines the concurrent training effect, discusses the potential molecular mechanisms underlying this effect, and proposes strategies to maximize strength and endurance in the high-level athlete. IntroductionRobert Hickson was a power-lifter when he went to do his post-doctoral work in the laboratory of Professor John Holloszy. Every day, Holloszy, the father of endurance exercise research, would leave the Washington University Medical Campus and go for runs through the adjoining Forest Park. In his effort to make a good impression with his new boss, Dr. Hickson decided to accompany Prof. Holloszy on his afternoon runs, but soon found that his muscle mass and strength were decreasing in spite of the fact that he was still doing his strength training at the same frequency and intensity. When Hickson approached Holloszy with his problem, he was told: “this should be the first study you do when you have your own lab.” True to his word, the first study that Hickson completed in his new laboratory at the University of Illinois in Chicago was the seminal study on concurrent training. Published in 1980 [], Hickson’s classic study trained three groups of subjects: Group 1 performed strength training alone; Group 2 performed endurance training alone; and Group 3 performed strength and endurance together. The strength training was performed 5 days per week for 10 weeks, and was designed exclusively to increase leg strength. True to his power-lifting background, Hickson had his subjects perform all of the exercises with as much weight as possible. The endurance training was performed 6 days per week for the same 10-week period and consisted of 3 days of cycling and 3 days of running. The cycling exercise consisted of six 5-min intervals at maximal aerobic capacity (VO2max), whereas the instructions on the running days were to “run as fast as possible” for 30 min/day in the first week, 35 min/day for the second week, and 40 min/day for the remainder of the study. The concurrent training group performed both the strength and endurance training protocols in a non-standardized order with between 15 min and 2 h of rest in between. At the end of the 10-week training program, VO2max was determined on the bike and treadmill. The strength alone group showed a 4 % improvement in VO2max on the bike with no change when measured on the treadmill. In contrast, the endurance and concurrent training groups both increased VO2max by 17 % on the treadmill and ~20 % on the bike. This indicated that strength training does not negatively affect endurance adaptations or performance. It should be noted, however, that the concurrent training group did not increase their bodyweight over the training period as a result of their strength training. If they had, it would be expected that their endurance performance could be affected, especially during running where they would have to support and propel this extra mass. As for strength, average strength in the concurrent and strength training groups increased at the same rate throughout the first 6–7 weeks of training (Fig. 1). Strength continued to increase throughout the entire 10-week training period in the strength training only group. In contrast, strength leveled off between the 7th and 8th weeks in the concurrent training group and surprisingly decreased during the 9th and 10th weeks of training. This indicates either that the concurrent training group was over-reaching or that high-intensity endurance exercise of a sufficient frequency can inhibit long-term strength adaptations. The concurrent training effect on strength. The figure shows the increase in one repetition maximum in the squat in subjects who participated in 10 weeks of high-intensity resistance exercise alone (resistance), endurance exercise alone (endurance), and both types of training (concurrent). Also, note that the strength and concurrent groups both increased their strength together up to 7 weeks, when the strength group started making greater gains than the concurrent group (adapted from Hickson [], with permission). 1RM one repetition maximum When others have repeated the frequency and intensity that Hickson employed in his study, they have found a similar attenuation in strength and, importantly, impaired muscle fiber hypertrophy [, ]. For example, Kraemer and colleagues [] showed that running and strength training at a high intensity for 4 days a week resulted in lower power concomitant with impaired muscle fiber hypertrophy than training for strength alone. Strength training alone resulted in ~28 % hypertrophy, whereas concurrent training resulted in only a ~16 % hypertrophy. This indicates that concurrent endurance training impairs not only strength but muscle hypertrophy as well. It is important to note, though, that when the frequency, intensity, or duration of training is decreased, the degree of interference decreases. For example, in two separate studies McCarthy and colleagues showed that cycling 3 days a week for 50 min at 70 % VO2max was not enough to impair strength [] or hypertrophy [] as a result of concurrent strength training. Further, Sillanpää et al. showed that cycling for 30 min twice a week below anaerobic threshold was not enough to impair strength or lean mass in middle-aged women [] or old men []. Taken together, these data suggest that strength and endurance increase concomitantly up to a point. However, once the frequency increases past 4 days a week or the intensity of endurance exercise increases above 80 % VO2max, endurance exercise prevents the increase in muscle mass and strength that occurs with strength training. This was illustrated nicely in a recent meta-analysis that demonstrated that the effect size of strength training alone on muscle hypertrophy was 1.22 and for strength was 1.71 []. The corresponding numbers for concurrent training were 0.8 and 1.28, indicating that, in a large cohort, endurance exercise impairs muscle size and strength adaptations []. Molecular Underpinning of Muscle HypertrophyIncreased strength is the combined effect of improvements in neural activation, muscle fiber size, and connective tissue stiffness. Therefore, concomitant endurance exercise could decrease adaptations of any/all of these physiological parameters. There does not appear to be a decrease in the neural (learning) adaptation since in the early stages of training, when the neural adaptation is the strongest (4, 6, and 8 weeks), strength is similar between strength and concurrent training groups [, ]. However, it is possible that neuromuscular fatigue plays a role in the decrease in force as training continues. At this point, no one has measured the effect of concurrent training on connective tissue stiffness, so we are unsure of the role of this tissue in the impaired strength response. In contrast, as stated above, there is some evidence that muscle hypertrophy is impaired in individuals training for both strength and endurance together compared with those training exclusively with strength exercises, and that this correlates quite well with the impaired strength response [, ]. Therefore, the primary effect of endurance exercise seems to be a decrease in resistance exercise-induced muscle hypertrophy. Over the last 15 years we have begun to understand the molecular events that lead to muscle hypertrophy and increased endurance capacity. These studies have shown that for exercise-induced muscle hypertrophy, the key signaling molecule is the mechanistic or mammalian target of rapamycin (mTOR). mTOR is a serine/threonine protein kinase that exists in two complexes. Both complexes contain the DEP domain-containing mTOR-interacting protein (DEPTOR) and the G-protein beta subunit-like protein (GβL; also known as lsT8). These proteins are negative and positive regulators of mTOR, respectively. Complex 1 (mTORC1) also contains the proline-rich Akt substrate of 40 kDa (PRAS40), an inhibitor of mTORC1 activity and the regulatory-associated protein of mTOR (raptor), which specifies the substrates that are phosphorylated by mTORC1. Raptor identifies the substrates for complex 1 by binding to TOS (TOR signaling) motifs, a five amino-acid sequence, found in proteins such as eukaryotic initiation factor (eIF) 4E binding protein-1 (4E-BP1), the 70-kDa ribosomal protein S6 kinase (S6K1), hypoxia-inducible factor-1 (HIF-1), and PRAS40. In contrast, complex 2 (mTORC2) contains the mammalian stress-activated map kinase-interacting protein 1 (mSIN1), which is important for targeting to membranes, the scaffold protein observed with rictor (PROTOR), and the rapamycin-insensitive companion of mTOR (rictor). Much like raptor in complex 1, rictor identifies the substrates that are phosphorylated by mTOR. However, rictor does not recognize TOS motifs and, as a result, in complex 2 mTOR is directed towards a completely different group of proteins including akt/PKB (protein kinase B), serum- and glucocorticoid-induced protein kinase (SGK), and protein kinase C (PKC). Importantly, the macrolide immunosuppressive antibiotic rapamycin specifically inhibits complex 1, allowing researchers to distinguish between the two complexes. Following resistance exercise there is a significant and sustained increase in the activity of mTORC1, as determined by an increase in S6K phosphorylation [] and activity []. The first indication that this increase in mTOR activity was important for resistance exercise-induced muscle hypertrophy came from work where we showed that the activity of mTOR 6 h following resistance exercise correlated with the increase in muscle mass following 6 weeks of training []. This finding in rats has since been demonstrated in humans [], suggesting that activation of mTORC1 is key to increasing muscle mass and strength. In many cells, mTOR is activated by growth factors as a way to stimulate protein synthesis []. However, resistance exercise activates mTOR in a growth factor-independent manner []. Unlike growth factors that use a receptor tyrosine kinase to signal through phosphoinositide 3-kinase (PI3K) to PKB, resistance exercise activates mTOR without activating PI3K []. Instead, resistance exercise activates an unidentified kinase (Fig. 2) that phosphorylates the potent mTOR inhibitor tuberin (TSC2) on RxRxx motifs []. When TSC2 is phosphorylated in this manner, it binds to 14-3-3 proteins and is moved away from mTOR and its activator Ras-homolog enriched in brain (Rheb). In this way, Rheb becomes activated and stimulates mTORC1 activity, leading to increased protein synthesis. The activation of the mechanistic target of rapamycin complex 1 (mTORC1, mTOR, and raptor complex) following resistance exercise and feeding. Lifting a heavy weight to failure stimulates a mechanoreceptor that in turn activates an RxRxxS*/T* kinase (depicted by ??? at the membrane) that phosphorylates and moves the TSC2 away from the lysosome allowing Rheb to remain in the GTP bound state. Simultaneously, amino acid uptake and intracellular amino acid levels increase. The extra amino acids stimulate the LRS to act as a GAP towards RagC/D and GATOR2 blocks GATOR1 (the GAP of RagA/B) and the Ragulator GTP loads RagA/B and activates the complex. The active Rag complex then binds to raptor and positions mTOR beside its activator: GTP bound Rheb. The resulting elevation of mTORC1 activity drives myofibrillar protein synthesis and eventually leads to an increase in muscle mass and strength. DEPTOR DEP (Dishevelled, Egl-10 and Pleckstrin) domain-containing mTOR-interacting protein, GβL G-protein beta subunit-like protein, GAP GTPase activating protein, GATOR GAP Activity Towards Rags, GDP guanosine diphosphate, GTP guanosine triphosphate, LAMP2 lysosome-associated membrane protein 2, LAT1 L-type amino acid transporter, LRS leucyl transfer RNA synthase, mTOR mammalian target of rapamycin, mTORC1 mTOR complex 1 P phosphorylation, PRAS40 proline-rich Akt substrate of 40 kDa, Rab7 Ras-related protein 7, raptor the regulatory-associated protein of mTOR, Rheb Ras homolog enriched in brain, TSC2 tublerosclerosis complex Contraction-induced dissociation of TSC2 from the lysosome is not the only thing that occurs following resistance exercise that leads to the prolonged activation of mTOR. In the hours after resistance exercise there is also an increase in the rate of amino acid uptake from the blood into the muscle. Specifically, leucine and glutamine are increased within the working muscle [, ]. This increase in leucine within the muscle is likely the result of an increase in the primary leucine transporter (LAT1) messenger RNA (mRNA) [] and protein []. Interestingly, the increase in glutamine can help drive leucine uptake since LAT1 transports glutamine out of the muscle as it transports leucine into the muscle in a process called tertiary active transport []. As leucine enters the muscle it acts to trigger protein synthesis largely through its ability to activate mTORC1 []. As leucine is taken up, it binds to the leucyl-transfer RNA (tRNA) synthetase (LRS). This not only conjugates leucine to its tRNAs, but the LRS also acts as the first step in the amino acid activation of the mTORC1 []. LRS may serve as a GTPase activating protein (GAP) towards the small G-protein (RagD), which in turn is a component of a heterodimer of RagA/B and RagC/D that is important for amino acid sensing. When RagD is bound to guanosine triphosphate (GTP) it forms an inactive complex. LRS catalyzes the hydrolysis of this GTP to guanosine diphosphate (GDP) and RagD then interacts with the Ragulator []. At the Ragulator, the Rag heterodimer interacts with GATOR (GAP Activity Towards Rags), an octomeric complex that controls the activity of the Rag proteins []. In the presence of amino acids, the Rags are activated and recruit mTORC1 to the lysosome by binding to raptor [, ]. As discussed above, the activator of mTOR, Rheb, is also located on the lysosome, so the net effect of leucine is to bring mTOR to its activator. Together, these data suggest that resistance exercise activates mTORC1 through the activation of an RxRxx-directed kinase that phosphorylates and moves the inhibitor TSC2 away from the lysosome (Fig. 2). At the same time, by regulating LAT1, enhanced amino acid uptake moves mTOR to the lysosome where it can be activated by GTP-bound Rheb. This complex molecular process explains both the load-dependent activation of mTOR (more activation of the RxRxx kinase []) and the effects of amino acid consumption (increased movement of mTOR to the lysosome and its activator Rheb [, ]) on protein synthesis and, finally, muscle hypertrophy [, ]. Even though a molecular mechanism exists for the activation of mTORC1 by resistance exercise and protein feeding, this does not prove that mTORC1 is what is necessary for muscle growth. Evidence that mTORC1 drives load-induced muscle hypertrophy was derived from experiments using rapamycin to specifically inhibit the kinase. In mice, Bodine and colleagues showed that daily injections of rapamycin could inhibit muscle hypertrophy [], and in humans, Drummond and colleagues showed that rapamycin could block the acute increase in protein synthesis after resistance exercise []. These experiments suggested that a rapamycin-sensitive process was required for load-induced muscle growth. The specific role of mTOR was demonstrated by an elegant study from the Hornberger laboratory using mice with a knock-in mutation that made mTOR resistant to treatment with rapamycin []. As would be expected, in the wild-type animals rapamycin completely blocked muscle hypertrophy. However, in the mice expressing a rapamycin-resistant mutant of mTOR muscle, hypertrophy occurred normally both in the presence and absence of rapamycin []. These data showed conclusively that resistance exercise-induced muscle hypertrophy is completely dependent on mTOR. Therefore, research looking for a molecular mechanism underlying the concurrent training effect has been focused exclusively on ways that endurance exercise could inhibit mTORC1 activity. Molecular Underpinning of Enhanced EnduranceWhereas the muscle hypertrophy response to resistance exercise appears to converge on a single protein complex (mTOR), endurance adaptations are the result a variety of metabolic signals and molecules. During endurance exercise the concentration of calcium, oxygen free radicals, adenosine monophosphate (AMP), lactate, nicotinamide adenine dinucleotide (NAD+), inorganic phosphate, and glycogen change dramatically within the working muscle. At the same time, systemic changes in hormones, such as catecholamines, may influence the muscle and drive adaptations. These metabolic and hormonal signals turn on signaling proteins within muscle that, if repeated at a sufficient frequency, lead to enhanced mitochondrial mass, improved fat and glucose oxidation, and increased capillary density. For example, calcium released during contraction activates the calcium-calmodulin kinase (CaMK) family of proteins, specifically CaMKII in skeletal muscle []. Active CaMK can increase both the capacity for glucose uptake through upregulation of the glucose transporter GLUT4 [], and mitochondrial mass by transcriptional upregulation of the mitochondrial biogenesis regulator PGC-1α (peroxisome proliferator γ coactivator 1α) []. The decrease in adenosine triphosphate (ATP) and glycogen and the rise in adenosine diphosphate (ADP) and AMP during high-intensity endurance exercise activates the AMP-activated protein kinase (AMPK). Active AMPK is involved in the increase in fat oxidation during exercise [] and also plays a role in the long-term regulation of mitochondrial mass by controlling the transcription and activity of PGC-1α []. The decrease in glycogen also activates the 38 kDa mitogen-activated protein kinase (p38), which, like AMPK, can increase the transcription and activity of PGC-1α [–]. The rise in lactate and NAD+ activates the NAD+-dependent deacetylase family of sirtuins (SIRT). Members of this family control metabolic flux through the tricarboxylic acid (TCA) cycle, insulin sensitivity [], and PGC-1α activity []. Last, the rise in circulating catecholamines through the β-adrenergic receptor activates the cyclic AMP response element binding protein (CREB), a transcription factor that is required for the transcriptional upregulation of PGC-1α []. Since all of these signaling molecules are activated by endurance exercise, it is possible that one or more of them can simultaneously inhibit mTOR activation and limit skeletal muscle hypertrophy during concurrent training. Beyond these signals, which are known to play a positive role in the adaptation to endurance exercise, the stress of exercise is known to increase other processes, such as free radical generation or endoplasmic reticulum (ER) stress [, ] that might influence mTOR activity or protein synthesis in response to resistance exercise. Current Data on the Molecular Underpinning of the Concurrent Training EffectThe first hint of a molecular mechanism that could explain how endurance exercise could impair muscle hypertrophy of concurrent strength training came when Inoki and colleagues showed that metabolic stress blocks mTORC1 activity []. Of keen interest for exercise physiologists was the fact that AMPK was required for the inhibitory effect of metabolic stress on mTOR []. The effect of metabolic stress on mTOR was first suggested to be the result of AMPK phosphorylating and activating the mTOR inhibitor TSC2 []. Later, another group showed that AMPK could phosphorylate raptor and dissociate the mTORC1 []. More recently, a third group has shown that in some cells, metabolic stress inhibits mTOR in an AMPK-independent manner by preventing mTOR localization to the lysosome []. Regardless of the mechanism, putting together the effect of metabolic stress/AMPK activation on mTOR and the fact that metabolic stress and AMPK activity were increased during endurance exercise, exercise physiologists began to ask the question “can AMPK limit muscle hypertrophy?” Thomson and Gordon were the first to show that impaired muscle growth was seen in rats where AMPK activity was higher, supporting the hypothesis that AMPK mediated the concurrent training effect []. They went further using the AMP mimetic AICAR (5-aminoimidazole-4-carboxamide ribonucleotide) to activate AMPK in muscles before resistance exercise and, consistent with the hypothesis, AICAR treatment blocked S6K phosphorylation []. We then used an animal model of concurrent training to show that the isoform of AMPK that was activated by concurrent training (α1) was not the form that was associated with endurance adaptations (α2), suggesting that during concurrent training a different form of AMPK was activated to prevent growth []. Consistent with the hypothesis that α1-AMPK limited growth, mice in which the α1 isoform of AMPK was knocked out showed a 33 % greater increase in muscle fiber size and enhanced mTOR signaling to S6K and 4E-BP1 in response to training than wild-type mice []. It is important to note that the α1-AMPK knockout mice showed significantly greater α2-AMPK activity in an effort to compensate for the loss of α1, but this was unable to restrict growth. Therefore, the metabolically activated α2 form of AMPK was not as effective at inhibiting mTOR as the α1 form. Concurrent Training Program TemplateEven though the animal studies have been impressive at showing that AMPK can directly inhibit mTORC1 activity and muscle growth, acute studies in humans are not as definitive. The most interesting of these studies is a pair from John Hawley’s laboratory [, ]. In the first, they showed that the activation of the mTORC1 following eight sets of five repetitions at 80 % of their one repetition maximum (1RM) was completely lost if the subjects had performed ten 6-second maximal sprint efforts on a bicycle 15 min before strength training, and mTOR activity rapidly returned to baseline if the sprint session was performed after strength training []. Interestingly, if, instead of using a high-intensity sprint session, the authors used a moderate intensity bout of cycling, there was no difference in mTORC1 activity []. Consistent with endurance exercise intensity being a key to the interference effect, Lundberg et al. did not find any inhibition of mTOR activation when subjects performed 45 min of cycling at 70 % VO2max 6 h before performing resistance exercise []. Further, Apró and colleagues did not report any decrease in mTOR signaling when subjects performed 30 min of cycling at 70 % of VO2max 15 min after completing a resistance training session []. These findings are completely consistent with the training data that show that the interference effect is only seen if the subjects train at a high frequency and intensity [, ], and the fact that the muscle AMP/ATP ratio and AMPK activity increases with exercise intensity []. Even though the intensity effects and the animal data are completely consistent with AMPK mediating the inhibition of mTOR activity during concurrent training, the activation of AMPK in both of the training groups was the same in the sprint interval study by Coffey and colleagues, suggesting that AMPK could not explain the inhibition of mTOR activity []. With the caveat that the phosphorylation of AMPK is not the most sensitive measure of AMPK activity (a direct measure of activity or the phosphorylation of its downstream target acetyl-CoA carboxylase show both the allosteric activation by AMP or ADP and the effect of phosphorylation []), this suggests that another molecular signal contributes to, or better explains, the inhibitory effect of endurance exercise on muscle hypertrophy. Alternative Molecular Underpinning of the Concurrent Training Effect
If the activation of AMPK does not completely explain the concurrent training effect, then what other molecular events are activated by endurance exercise that could block mTOR and/or inhibit muscle hypertrophy? As discussed in Sect. 3, endurance exercise activates the sirtuin family of NAD+-dependent deactetylases including SIRT1 []. Like AMPK, SIRT1 is activated by metabolic stress and as a result of its relationship with lactate/NAD+, is activated in an intensity-dependent manner []. Further, SIRT1 is able to inhibit mTOR []. In HeLa cells, where AMPK activity is reduced, the knockdown or inhibition of SIRT1 increased mTORC1 activity, whereas the SIRT1 activator resveratrol decreased mTOR activity []. Further, since SIRT1 and AMPK signaling are closely linked [], it is possible that SIRT1 and not AMPK is the direct mediator of mTOR inactivation that was discussed above following high-intensity exercise. 
Another way that endurance exercise could inhibit mTORC1 activity is through the unfolded protein, or ER stress, pathway. Periods of high lipid exposure, glucose deprivation, or increased synthesis of secretory proteins, lead to the accumulation of unfolded or misfolded proteins within the ER lumen []. To cope with the increase in unfolded proteins, cells activate the unfolded protein response, a series of events that serve to block general protein synthesis, increase protein-folding capacity, and restore ER function. Interestingly, the unfolded protein response is activated in muscle by acute endurance exercise [], a high-fat diet [], or the combination of both stimuli []. Furthermore, both endurance exercise [] and a high-fat diet [] impair muscle hypertrophy and ER stress decreases mTORC1 activity and protein synthesis in muscle [, ]. Therefore, like AMPK and SIRT1, the ER stress response could contribute to the concurrent training effect. Science-Based Recommendations for Training to Maximize Concurrent TrainingUsing the molecular information provided in Sects. 2–5, some simple nutritional and training strategies can be devised to maximize the adaptations to concurrent training. The goal of these recommendations is to maximize the mitochondrial adaptation to endurance exercise and the muscle mass and strength adaptation to strength training. To do this, the following could be recommended:
ConclusionsThese simple recommendations, based on our current understanding of the molecular response to exercise, should allow for the maximal adaptive response to both endurance and strength exercise. However, it is important to remember that what makes a good molecular biologist is the ability to break down complex physiological processes into simple molecular switches. Naturally, improving endurance and strength together in an elite athlete is more than just striking the balance between AMPK/SIRT1 and mTORC1. This is especially true in situations where performance is based on skill optimization that goes well beyond these simple molecular pathways. In the end, how an athlete performs with their improved endurance and strength is based on far more complex processes that are unfortunately poorly understood. AcknowledgmentsThis article was published in a supplement supported by the Gatorade Sports Science Institute (GSSI). The supplement was guest edited by Lawrence L. Spriet who attended a meeting of the GSSI Expert Panel in February 2013 and received honoraria from the GSSI, a division of PepsiCo, Inc., for his meeting participation and the writing of his manuscript. He has not received any honoraria for guest editing the supplement. Lawrence L. Spriet selected peer reviewers for each paper and managed the process. Keith Baar, PhD attended a meeting of the GSSI Expert Panel in February 2013 and received honoraria from the GSSI, a division of PepsiCo, Inc., for his meeting participation and the writing of this manuscript. The views expressed in this manuscript are those of the author and do not necessarily reflect the position or policy of PepsiCo, Inc. The author thanks Daniel West for critical review of the manuscript. References
1. Hickson RC. Interference of strength development by simultaneously training for strength and endurance. Eur J Appl Physiol Occup Physiol. 1980;45:255–263. doi: 10.1007/BF00421333. [PubMed] [CrossRef] [Google Scholar]
2. Kraemer WJ, Patton JF, Gordon SE, et al. Compatibility of high-intensity strength and endurance training on hormonal and skeletal muscle adaptations. J Appl Physiol. 1995;78:976–989. [PubMed] [Google Scholar]
3. Wilson JM, Marin PJ, Rhea MR, et al. Concurrent training: a meta-analysis examining interference of aerobic and resistance exercises. J Strength Cond Res. 2012;26:2293–2307. doi: 10.1519/JSC.0b013e31823a3e2d. [PubMed] [CrossRef] [Google Scholar]
4. McCarthy JP, Agre JC, Graf BK, et al. Compatibility of adaptive responses with combining strength and endurance training. Med Sci Sports Exerc. 1995;27:429–436. doi: 10.1249/00005768-199503000-00021. [PubMed] [CrossRef] [Google Scholar]
5. McCarthy JP, Pozniak MA, Agre JC. Neuromuscular adaptations to concurrent strength and endurance training. Med Sci Sports Exerc. 2002;34:511–519. doi: 10.1097/00005768-200203000-00019. [PubMed] [CrossRef] [Google Scholar]
6. Sillanpää E, Laaksonen DE, Häkkinen A, et al. Body composition, fitness, and metabolic health during strength and endurance training and their combination in middle-aged and older women. Eur J Appl Physiol. 2009;106:285–296. doi: 10.1007/s00421-009-1013-x. [PubMed] [CrossRef] [Google Scholar]
7. Sillanpää E, Häkkinen A, Nyman K, et al. Body composition and fitness during strength and/or endurance training in older men. Med Sci Sports Exerc. 2008;40:950–958. doi: 10.1249/MSS.0b013e318165c854. [PubMed] [CrossRef] [Google Scholar]
8. Baar K, Esser K. Phosphorylation of p70(S6k) correlates with increased skeletal muscle mass following resistance exercise. Am J Physiol Cell Physiol. 1999;276:C120–C127. [PubMed] [Google Scholar]
9. MacKenzie MG, Hamilton DL, Murray JT, et al. mVps34 is activated following high-resistance contractions. J Physiol. 2009;587:253–260. doi: 10.1113/jphysiol.2008.159830.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
10. Terzis G, Georgiadis G, Stratakos G, et al. Resistance exercise-induced increase in muscle mass correlates with p70S6 kinase phosphorylation in human subjects. Eur J Appl Physiol. 2008;102:145–152. doi: 10.1007/s00421-007-0564-y. [PubMed] [CrossRef] [Google Scholar]
11. Sengupta S, Peterson TR, Sabatini DM. Regulation of the mTOR complex 1 pathway by nutrients, growth factors, and stress. Mol Cell. 2010;40:310–322. doi: 10.1016/j.molcel.2010.09.026.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
12. Philp A, Hamilton DL, Baar K. Signals mediating skeletal muscle remodeling by resistance exercise: PI3-kinase independent activation of mTORC1. J Appl Physiol. 2011;110:561–568. doi: 10.1152/japplphysiol.00941.2010. [PubMed] [CrossRef] [Google Scholar]
13. Hamilton DL, Philp A, MacKenzie MG, et al. A limited role for PI(3,4,5)P3 regulation in controlling skeletal muscle mass in response to resistance exercise. PLoS One. 2010;5:e11624. doi: 10.1371/journal.pone.0011624.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
14. Jacobs BL, You JS, Frey JW, et al. Eccentric contractions increase the phosphorylation of tuberous sclerosis complex-2 (TSC2) and alter the targeting of TSC2 and the mechanistic target of rapamycin to the lysosome. J Physiol. 2013;591:4611–4620. doi: 10.1113/jphysiol.2013.256339.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
15. Biolo G, Maggi SP, Williams BD, et al. Increased rates of muscle protein turnover and amino acid transport after resistance exercise in humans. Am J Physiol Endocrinol Metab. 1995;268:E514–E520. [PubMed] [Google Scholar]
16. Churchward-Venne TA, Burd NA, Mitchell CJ, et al. Supplementation of a suboptimal protein dose with leucine or essential amino acids: effects on myofibrillar protein synthesis at rest and following resistance exercise in men. J Physiol. 2012;590:2751–2765. doi: 10.1113/jphysiol.2012.228833.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
17. Drummond MJ, Fry CS, Glynn EL, et al. Skeletal muscle amino acid transporter expression is increased in young and older adults following resistance exercise. J Appl Physiol. 2011;111:135–142. doi: 10.1152/japplphysiol.01408.2010.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
18. Baird FE, Bett KJ, MacLean C, et al. Tertiary active transport of amino acids reconstituted by coexpression of System A and L transporters in Xenopus oocytes. Am J Physiol Endocrinol Metab. 2009;297:E822–E829. doi: 10.1152/ajpendo.00330.2009. [PubMed] [CrossRef] [Google Scholar]
19. Crozier SJ, Kimball SR, Emmert SW, et al. Oral leucine administration stimulates protein synthesis in rat skeletal muscle. J Nutr. 2005;135:376–382. [PubMed] [Google Scholar]
20. Han JM, Jeong SJ, Park MC, et al. Leucyl-tRNA synthetase is an intracellular leucine sensor for the mTORC1-signaling pathway. Cell. 2012;149:410–424. doi: 10.1016/j.cell.2012.02.044. [PubMed] [CrossRef] [Google Scholar]
21. Sancak Y, Bar-Peled L, Zoncu R, et al. Ragulator-Rag complex targets mTORC1 to the lysosomal surface and is necessary for its activation by amino acids. Cell. 2010;141:290–303. doi: 10.1016/j.cell.2010.02.024.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
22. Bar-Peled L, Chantranupong L, Cherniack AD, et al. A tumor suppressor complex with GAP activity for the Rag GTPases that signal amino acid sufficiency to mTORC1. Science. 2013;340:1100–1106. doi: 10.1126/science.1232044.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
23. Sancak Y, Peterson TR, Shaul YD, et al. The Rag GTPases bind raptor and mediate amino acid signaling to mTORC1. Science. 2008;320:1496–1501. doi: 10.1126/science.1157535.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
24. Kim E, Goraksha-Hicks P, Li L, et al. Regulation of TORC1 by Rag GTPases in nutrient response. Nat Cell Biol. 2008;10:935–945. doi: 10.1038/ncb1753.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
25. Tipton KD, Ferrando AA, Phillips SM, et al. Postexercise net protein synthesis in human muscle from orally administered amino acids. Am J Physiol Endocrinol Metab. 1999;276:E628–E634. [PubMed] [Google Scholar]
26. Hartman JW, Tang JE, Wilkinson SB, et al. Consumption of fat-free fluid milk after resistance exercise promotes greater lean mass accretion than does consumption of soy or carbohydrate in young, novice, male weightlifters. Am J Clin Nutr. 2007;86:373–381. [PubMed] [Google Scholar]
27. Bodine SC, Stitt TN, Gonzalez M, et al. Akt/mTOR pathway is a crucial regulator of skeletal muscle hypertrophy and can prevent muscle atrophy in vivo. Nat Cell Biol. 2001;3:1014–1019. doi: 10.1038/ncb1101-1014. [PubMed] [CrossRef] [Google Scholar]
28. Drummond MJ, Fry CS, Glynn EL, et al. Rapamycin administration in humans blocks the contraction-induced increase in skeletal muscle protein synthesis. J Physiol. 2009;587:1535–1546. doi: 10.1113/jphysiol.2008.163816.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
29. Goodman CA, Frey JW, Mabrey DM, et al. The role of skeletal muscle mTOR in the regulation of mechanical load-induced growth. J Physiol. 2011;589:5485–5501.[PMC free article] [PubMed] [Google Scholar]
30. Rose AJ, Kiens B, Richter EA. Ca2+-calmodulin-dependent protein kinase expression and signalling in skeletal muscle during exercise. J Physiol. 2006;574:889–903. doi: 10.1113/jphysiol.2006.111757.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
31. Smith JA, Kohn TA, Chetty AK, et al. CaMK activation during exercise is required for histone hyperacetylation and MEF2A binding at the MEF2 site on the Glut4 gene. Am J Physiol Endocrinol Metab. 2008;295:E698–E704. doi: 10.1152/ajpendo.00747.2007. [PubMed] [CrossRef] [Google Scholar]
32. Wu H, Kanatous SB, Thurmond FA, et al. Regulation of mitochondrial biogenesis in skeletal muscle by CaMK. Science. 2002;296:349–352. doi: 10.1126/science.1071163. [PubMed] [CrossRef] [Google Scholar]
33. Winder WW, Hardie DG. Inactivation of acetyl-CoA carboxylase and activation of AMP-activated protein kinase in muscle during exercise. Am J Physiol Endocrinol Metab. 1996;270:E299–E304. [PubMed] [Google Scholar]
34. Hardie DG, Ross FA, Hawley SA. AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat Rev Mol Cell Biol. 2012;13:251–262. doi: 10.1038/nrm3311.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
35. Akimoto T, Pohnert SC, Li P, et al. Exercise stimulates PGC-1alpha transcription in skeletal muscle through activation of the p38 MAPK pathway. J Biol Chem. 2005;280:19587–19593. doi: 10.1074/jbc.M408862200. [PubMed] [CrossRef] [Google Scholar]
36. Pogozelski AR, Geng T, Li P, et al. p38gamma mitogen-activated protein kinase is a key regulator in skeletal muscle metabolic adaptation in mice. PLoS One. 2009;4:e7934. doi: 10.1371/journal.pone.0007934.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
37. Puigserver P, Rhee J, Lin J, et al. Cytokine stimulation of energy expenditure through p38 MAP kinase activation of PPARgamma coactivator-1. Mol Cell. 2001;8:971–982. doi: 10.1016/S1097-2765(01)00390-2. [PubMed] [CrossRef] [Google Scholar]
38. Schenk S, McCurdy CE, Philp A, et al. Sirt1 enhances skeletal muscle insulin sensitivity in mice during caloric restriction. J Clin Invest. 2011;121:4281–4288. doi: 10.1172/JCI58554.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
39. Rodgers JT, Lerin C, Haas W, et al. Nutrient control of glucose homeostasis through a complex of PGC-1alpha and SIRT1. Nature. 2005;434:113–118. doi: 10.1038/nature03354. [PubMed] [CrossRef] [Google Scholar]
40. Chinsomboon J, Ruas J, Gupta RK, et al. The transcriptional coactivator PGC-1alpha mediates exercise-induced angiogenesis in skeletal muscle. Proc Natl Acad Sci U S A. 2009;106:21401–21406. doi: 10.1073/pnas.0909131106.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
41. Deldicque L, Cani PD, Delzenne NM, et al. Endurance training in mice increases the unfolded protein response induced by a high-fat diet. J Physiol Biochem. 2013;69:215–225. doi: 10.1007/s13105-012-0204-9. [PubMed] [CrossRef] [Google Scholar]
42. Wu J, Ruas JL, Estall JL, et al. The unfolded protein response mediates adaptation to exercise in skeletal muscle through a PGC-1alpha/ATF6alpha complex. Cell Metab. 2011;13:160–169. doi: 10.1016/j.cmet.2011.01.003.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
43. Inoki K, Zhu T, Guan KL. TSC2 mediates cellular energy response to control cell growth and survival. Cell. 2003;115:577–590. doi: 10.1016/S0092-8674(03)00929-2. [PubMed] [CrossRef] [Google Scholar]
44. Gwinn DM, Shackelford DB, Egan DF, et al. AMPK phosphorylation of raptor mediates a metabolic checkpoint. Mol Cell. 2008;30:214–226. doi: 10.1016/j.molcel.2008.03.003.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
45. Kim SG, Hoffman GR, Poulogiannis G, et al. Metabolic stress controls mTORC1 lysosomal localization and dimerization by regulating the TTT-RUVBL1/2 complex. Mol Cell. 2013;49:172–185.[PMC free article] [PubMed] [Google Scholar]
46. Thomson DM, Gordon SE. Diminished overload-induced hypertrophy in aged fast-twitch skeletal muscle is associated with AMPK hyperphosphorylation. J Appl Physiol. 2005;98:557–564. doi: 10.1152/japplphysiol.00811.2004. [PubMed] [CrossRef] [Google Scholar]
47. Thomson DM, Fick CA, Gordon SE. AMPK activation attenuates S6K1, 4E-BP1, and eEF2 signaling responses to high-frequency electrically stimulated skeletal muscle contractions. J Appl Physiol. 2008;104:625–632. doi: 10.1152/japplphysiol.00915.2007. [PubMed] [CrossRef] [Google Scholar]
48. McGee SL, Mustard KJ, Hardie DG, et al. Normal hypertrophy accompanied by phosphoryation and activation of AMP-activated protein kinase alpha1 following overload in LKB1 knockout mice. J Physiol. 2008;586:1731–1741. doi: 10.1113/jphysiol.2007.143685.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
49. Mounier R, Lantier L, Leclerc J, et al. Important role for AMPKalpha1 in limiting skeletal muscle cell hypertrophy. Faseb J. 2009;23:2264–2273. doi: 10.1096/fj.08-119057. [PubMed] [CrossRef] [Google Scholar]
50. Coffey VG, Jemiolo B, Edge J, et al. Effect of consecutive repeated sprint and resistance exercise bouts on acute adaptive responses in human skeletal muscle. Am J Physiol Reg Integr Comp Physiol. 2009;297:R1441–R1451. doi: 10.1152/ajpregu.00351.2009. [PubMed] [CrossRef] [Google Scholar]
51. Coffey VG, Pilegaard H, Garnham AP, et al. Consecutive bouts of diverse contractile activity alter acute responses in human skeletal muscle. J Appl Physiol. 2009;106:1187–1197. doi: 10.1152/japplphysiol.91221.2008. [PubMed] [CrossRef] [Google Scholar]
52. Lundberg TR, Fernandez-Gonzalo R, Gustafsson T, et al. Aerobic exercise alters skeletal muscle molecular responses to resistance exercise. Med Sci Sports Exerc. 2012;44:1680–1688. doi: 10.1249/MSS.0b013e318256fbe8. [PubMed] [CrossRef] [Google Scholar]
53. Apró W, Wang L, Ponten M, et al. Resistance exercise induced mTORC1 signaling is not impaired by subsequent endurance exercise in human skeletal muscle. Am J Physiol Endocrinol Metab. 2013;305:E22–E32. doi: 10.1152/ajpendo.00091.2013. [PubMed] [CrossRef] [Google Scholar]
54. Chen ZP, Stephens TJ, Murthy S, et al. Effect of exercise intensity on skeletal muscle AMPK signaling in humans. Diabetes. 2003;52:2205–2212. doi: 10.2337/diabetes.52.9.2205. [PubMed] [CrossRef] [Google Scholar]
55. Philp A, Chen A, Lan D, et al. Sirtuin 1 (SIRT1) deacetylase activity is not required for mitochondrial biogenesis or peroxisome proliferator-activated receptor-gamma coactivator-1alpha (PGC-1alpha) deacetylation following endurance exercise. J Biol Chem. 2011;286:30561–30570. doi: 10.1074/jbc.M111.261685.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
56. Philp A, Schenk S. Unraveling the complexities of SIRT1-mediated mitochondrial regulation in skeletal muscle. Exerc Sport Sci Rev. 2013;41:174–181. doi: 10.1097/JES.0b013e3182956803.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
57. Ghosh HS, McBurney M, Robbins PD. SIRT1 negatively regulates the mammalian target of rapamycin. PLoS One. 2010;5:e9199. doi: 10.1371/journal.pone.0009199.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
58. Canto C, Jiang LQ, Deshmukh AS, et al. Interdependence of AMPK and SIRT1 for metabolic adaptation to fasting and exercise in skeletal muscle. Cell Metab. 2010;11:213–219. doi: 10.1016/j.cmet.2010.02.006.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
59. Zhang K, Kaufman RJ. Protein folding in the endoplasmic reticulum and the unfolded protein response. Handb Exp Pharmacol. 2006;(172):69–91. [PubMed]
60. Deldicque L, Cani PD, Philp A, et al. The unfolded protein response is activated in skeletal muscle by high-fat feeding: potential role in the downregulation of protein synthesis. Am J Physiol Endocrinol Metab. 2010;299:E695–E705. doi: 10.1152/ajpendo.00038.2010. [PubMed] [CrossRef] [Google Scholar]
61. Sitnick M, Bodine SC, Rutledge JC. Chronic high fat feeding attenuates load-induced hypertrophy in mice. J Physiol. 2009;587:5753–5765. doi: 10.1113/jphysiol.2009.180174.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
62. Deldicque L, Bertrand L, Patton A, et al. ER stress induces anabolic resistance in muscle cells through PKB-induced blockade of mTORC1. PLoS One. 2011;6:e20993. doi: 10.1371/journal.pone.0020993.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
63. Wojtaszewski JF, Nielsen P, Hansen BF, et al. Isoform-specific and exercise intensity-dependent activation of 5’-AMP-activated protein kinase in human skeletal muscle. J Physiol. 2000;528:221–226. doi: 10.1111/j.1469-7793.2000.t01-1-00221.x.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
64. Areta JL, Burke LM, Ross ML, et al. Timing and distribution of protein ingestion during prolonged recovery from resistance exercise alters myofibrillar protein synthesis. J Physiol. 2013;591:2319–2331.[PMC free article] [PubMed] [Google Scholar]
65. Res PT, Groen B, Pennings B, et al. Protein ingestion before sleep improves postexercise overnight recovery. Med Sci Sports Exerc. 2012;44:1560–1569. doi: 10.1249/MSS.0b013e31824cc363. [PubMed] [CrossRef] [Google Scholar]
66. McBride A, Ghilagaber S, Nikolaev A, et al. The glycogen-binding domain on the AMPK beta subunit allows the kinase to act as a glycogen sensor. Cell Metab. 2009;9:23–34. doi: 10.1016/j.cmet.2008.11.008.[PMC free article] [PubMed] [CrossRef] [Google Scholar]
67. Mettler S, Mitchell N, Tipton KD. Increased protein intake reduces lean body mass loss during weight loss in athletes. Med Sci Sports Exerc. 2010;42:326–337. doi: 10.1249/MSS.0b013e3181b2ef8e. [PubMed] [CrossRef] [Google Scholar]
68. Wang L, Mascher H, Psilander N, et al. Resistance exercise enhances the molecular signaling of mitochondrial biogenesis induced by endurance exercise in human skeletal muscle. J Appl Physiol. 2011;111:1335–1344. doi: 10.1152/japplphysiol.00086.2011. [PubMed] [CrossRef] [Google Scholar]
Comments are closed.
|
- Blog
- Home
- New Page
- Ufix 2
- Ram Leela Full Movie Download
- Tarzan Full Movie 2016
- Ek Rishta Songs
- Tp Link 722
- The Avengers Full Movie In Hindi Download
- Best Vst Crack Sites
- Wiro Sableng 2018 Download
- Birdman Gift To Toni Braxton
- 2018 Tamil Movies Download
- Download Sd Gundam Capsule Fighter
- Sony Vegas 12 Download Free
- Saint Seiya Asgard Full Episodes
- Novel Romantis Dewasa
- Foxit Software Review
- Etv Serials In Telugu
- Dave And Busters Play Cards
- Solidworks 2014 Free Download Crack
- The Karate Kid Full Movie 2010 Free
- Bhag Milkha Bhag Movie
- Anne Of Green Gables 2018
- The License Authorization Wizard
- Warriors Orochi 4
- Cheap Cigarettes Online Free Shipping
- The Exorcist Full Movie
- What Is My Windows 10 Serial Number
- Rihanna Cheers Youtube
- Mobile Suit Gundam Online Mmorpg
- Sketchup Pro Free License
- Download Windows Server 2008 R2 Iso Image
- Download Film Boruto Subtitle Indonesia
- Telangana Voter Id Application Status Check
- Alat Pemadam Kebakaran
- Totally Free Home Bookkeeping Software
- Camfrog Video Chat Room
- Download Mirrorop Sender
- Beyond Compare 4 Crack Keygen
- Hugh Glass True Story

 RSS Feed
RSS Feed
